ARS Pharma Shares Plummet After FDA Rejects Needle-Free Allergy Spray
Regulator's surprise decision on neffy nasal spray prompts significant stock decline and delays market entry.

UGANDA —
Key facts
- ARS Pharmaceuticals' stock fell sharply after the FDA declined to approve its neffy nasal spray.
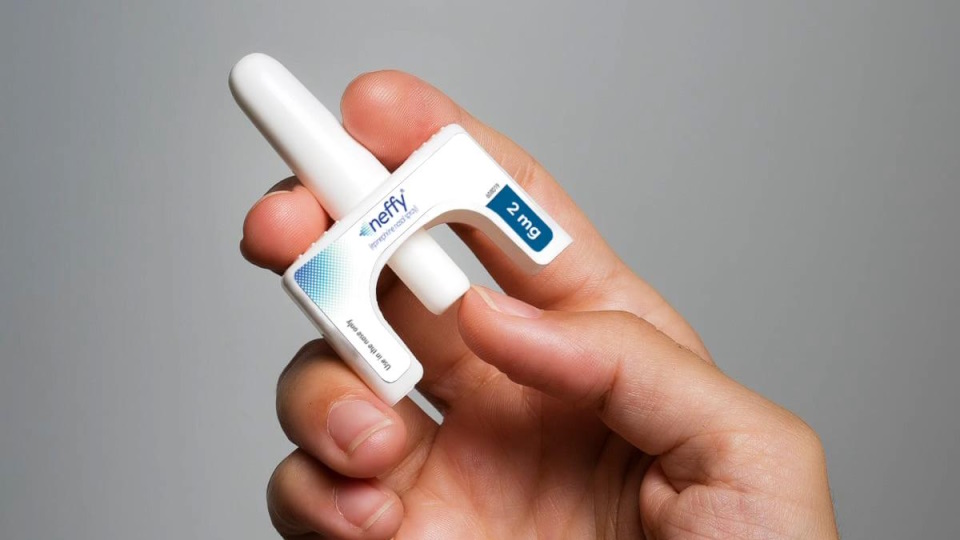
- Neffy is designed as a needle-free alternative to epinephrine injections for severe allergic reactions.
- An FDA advisory committee had recommended neffy's approval in May with a 16-to-six vote.
- The FDA issued a complete response letter (CRL) requesting results from a repeat-dose study.
- ARS Pharma aims to refile for approval in the first half of 2024, targeting year-end launch.
- The company held $252.5 million in cash reserves at the end of the second quarter.
- Viatris' EpiPen currently dominates the anaphylaxis emergency medication market.
FDA Decision Halts Promising Allergy Treatment
Shares in ARS Pharmaceuticals experienced a dramatic drop in pre-market trading following the U.S. Food and Drug Administration's (FDA) decision to reject the company's application for its novel epinephrine nasal spray, neffy. The drug, intended to be the first needle-free option for treating severe allergic reactions, faced a setback after the regulatory agency issued a complete response letter (CRL). The CRL signifies that the FDA has not approved the drug in its current form, a move that has sent shockwaves through the company and its investors. This unexpected turn of events jeopardizes the anticipated market entry of a product designed to offer a less intimidating alternative to traditional epinephrine auto-injectors. The implications of this rejection are significant, potentially delaying the availability of a much-needed treatment option for millions at risk of anaphylaxis.
Neffy's Promise and the FDA's Stance
Neffy was developed as a nasal spray to provide an alternative to epinephrine injections like Kaléo's AUVI-Q and Viatris' EpiPen, which currently hold a dominant position in the market for emergency anaphylaxis treatment. The spray was filed for approval for use in adults and children weighing 30 mg or more. ARS Pharmaceuticals expressed considerable surprise at the FDA's decision, particularly given that an advisory committee had recommended approval in May with a substantial 16-to-six vote, without requiring additional trials. The company also stated it had reached an agreement with the FDA in August concerning physician labeling and post-market requirements, which included a repeat-dose study. However, the FDA now appears to require the results of this specific repeat-dose study before granting approval, a condition that could push the program's timeline back by several months.
Market Reaction and Company Outlook
The news of the FDA's CRL triggered a sharp decline in ARS Pharma's stock. The company's shares lost 11% of their value late yesterday and plunged an additional 47% by the time of writing. This significant market reaction underscores the high expectations placed on neffy. Richard Lowenthal, co-founder and chief executive of ARS Pharma, articulated the company's strong belief in neffy's potential benefits for patients, families, and caregivers. He highlighted its potential to be more easily carried and administered, thereby reducing anxiety and hesitation during critical moments. Despite the disappointment, Lowenthal confirmed that the company's substantial cash reserves of $252.5 million at the close of the second quarter are sufficient to fund the additional study. ARS Pharma anticipates having an estimated $195 million remaining upon neffy's eventual launch, assuming it proceeds on the revised timeline.
Path to Refiling and Market Competition
ARS Pharmaceuticals is now working towards refiling its application for approval in the first half of 2024. The company expressed hope for receiving regulatory clearance before the end of that year. The delay comes at a time when the market for epinephrine auto-injectors is already subject to scrutiny. Earlier this year, the U.S. Supreme Court declined to hear a legal challenge against Viatris, which had accused the company of monopolizing the epinephrine auto-injector market through exclusive formulary agreements. The competitive landscape means that any delay in neffy's approval could allow established players to further solidify their market share, making the eventual launch even more critical for ARS Pharmaceuticals.
Broader Context of Allergy Treatment
Severe allergic reactions, or anaphylaxis, represent a significant public health concern, necessitating rapid and effective intervention. Epinephrine auto-injectors have long been the standard of care, but their needle-based delivery system can be a source of anxiety for users and caregivers. The development of a needle-free alternative like neffy has been keenly anticipated by patient advocacy groups and medical professionals alike. The ease of administration and reduced psychological barrier associated with a nasal spray could fundamentally change how individuals manage their risk of anaphylaxis. This FDA decision, while a setback for ARS Pharma, also highlights the rigorous standards regulatory bodies uphold to ensure the safety and efficacy of new medical treatments, particularly those intended for life-threatening conditions.
The bottom line
- ARS Pharmaceuticals' stock value significantly decreased following the FDA's rejection of its neffy nasal spray.
- Neffy aimed to be the first needle-free epinephrine nasal spray for severe allergic reactions, offering an alternative to injections.
- The FDA's complete response letter cited the need for results from a repeat-dose study before approval.
- Despite the setback, ARS Pharma plans to refile for approval in early to mid-2024, with a target launch by year-end.
- The company possesses substantial cash reserves to fund the required additional study.
- The established epinephrine auto-injector market, dominated by Viatris' EpiPen, faces ongoing legal and competitive pressures.




Richarlison Header Lifts Tottenham Out of Drop Zone as De Zerbi Revives Sinking Spurs

Roma Crush Fiorentina 4-0 as Lazio Edge Cremonese 2-1 to Reignite European Hopes
Noslin and Isaksen Lead Lazio Comeback to Beat Cremonese 2-1
